See the long-term exploratory analyses for BAVENCIO
SEE 5+ YEARS OF EFFICACY FOR BAVENCIO1
Don't miss the real-world evidence below
- Primary and Long-Term Analyses
- Long-Term Exploratory Analyses
- Patient-Reported Outcomes
Strategize for Extended Overall Survival with BAVENCIO 1L Maintenance2,3
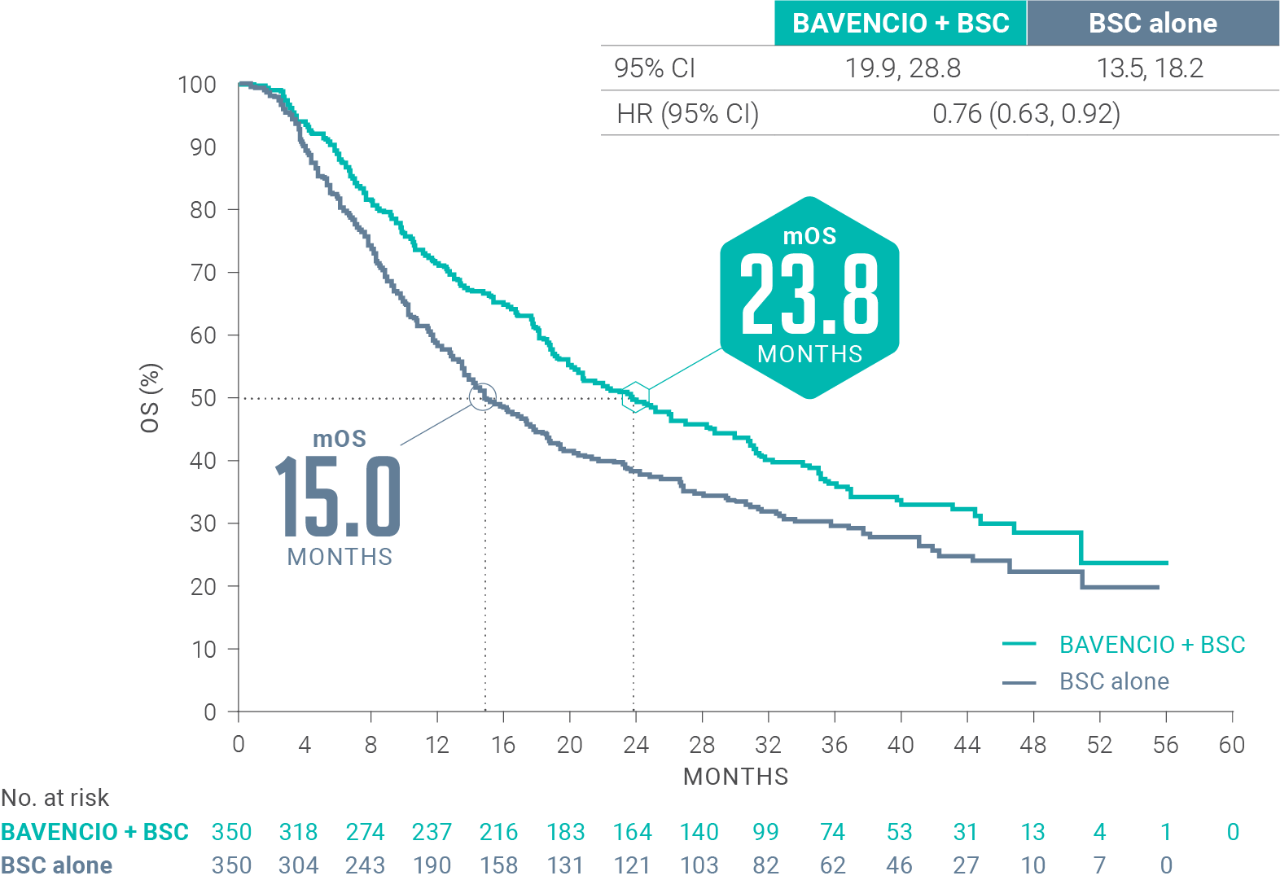
JAVELIN BLADDER 100 PRIMARY ANALYSIS: BAVENCIO + BSC demonstrated superior OS vs BSC alone2
Median follow-up: 19.6 months (95% CI: 18.0, 20.6) in the BAVENCIO + BSC arm; 19.2 months (95% CI: 17.4, 21.6) in the BSC-alone arm4
mOS of 21.4 MONTHS (95% CI: 18.9, 26.1) with BAVENCIO + BSC vs 14.3 MONTHS (95% CI: 12.9, 17.9) with BSC alone (n=350 in each arm); HR 0.69 (0.56, 0.86); 2-sided P valuea=0.0012
The pre-planned interim analysis was considered the primary analysis since the primary endpoint was met.3,5
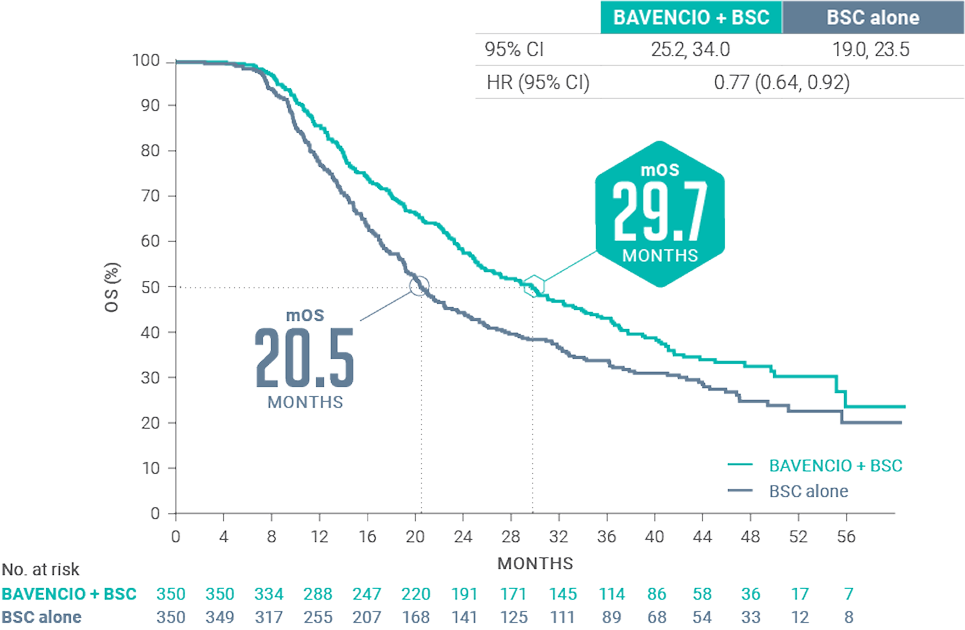
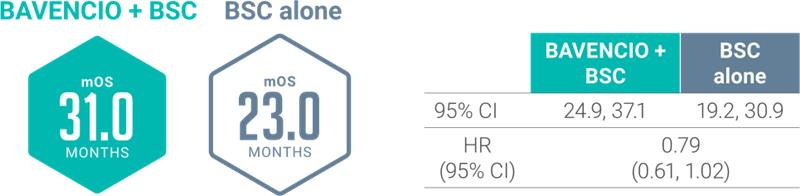
LONG-TERM ANALYSIS (3+ years): Consistent OS results were observed2
Median follow-up: 38.0 months (95% CI: 36.1, 40.5) in the BAVENCIO + BSC arm; 39.6 months (95% CI: 36.2, 41.7) in the BSC-alone arm3,5
| BAVENCIO + BSC | BSC alone | |
|---|---|---|
| 95% Cl | 19.9, 28.8 | 13.5, 18.2 |
| HR (95% Cl) | 0.76 (0.63, 0.92) | |
Long-term OS results in PD-L1–positive patients2,b (n=358, 51% of patients): HR 0.69 (95% CI: 0.52, 0.90)
PD-L1–negative tumors2 (exploratory analysis; n=270, 39% of patients): OS HR 0.82 (95% CI: 0.62, 1.09)
While the long-term OS analysis was prespecified, no formal hypothesis testing was performed given OS was met in the interim analysis. No conclusions can be drawn from this analysis.
aP value based on stratified log-rank.6
bUsing the VENTANA PD-L1 (SP263) assay, PD-L1–positive status was defined as PD-L1 expression in ≥25% of tumor cells or in ≥25% or 100% of tumor-associated immune cells if the percentage of immune cells was >1% or ≤1%, respectively. If none of these criteria were met, PD-L1 status was considered negative.6
Additional exploratory data from the JAVELIN Bladder 100 trial
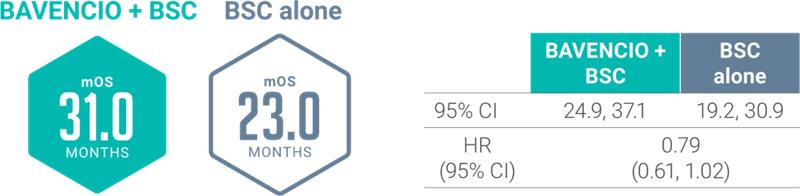
mOS of ~30 months from start of 1L platinum-containing chemotherapy5
LIMITATIONS:
These are exploratory, post hoc analyses of OS data, inclusive of platinum-containing chemotherapy (4-6 cycles), treatment-free interval (4-10 weeks, per trial protocol), randomized study treatment with BAVENCIO + BSC or BSC alone, and subsequent therapy. This analysis includes only patients who did not progress on first-line platinum-containing chemotherapy and subsequently enrolled in the JAVELIN Bladder 100 trial. Small patient numbers can be a limitation of subgroup analyses. Safety data are not available pre-randomization. No conclusions can be drawn from these OS analyses.
JB100 SUBGROUP ANALYSES showed mOS from the start of 1L platinum-containing chemotherapy regardless of type5
Prespecified Analysis: Cisplatin + Gemcitabinea
Prespecified Analysis: Carboplatin + Gemcitabinea

aStratified HR analyses are shown.7
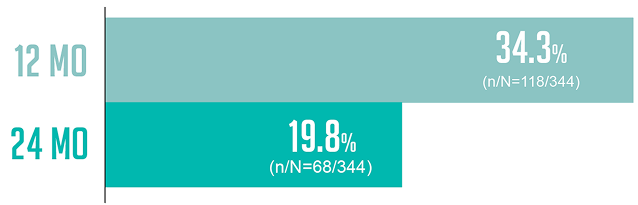
AN EXPLORATORY, POST HOC ANALYSIS: Duration of Treatment
An exploratory, post hoc analysis of patients who received BAVENCIO + BSC8
An exploratory, post hoc analysis of patients who received BAVENCIO + BSC8
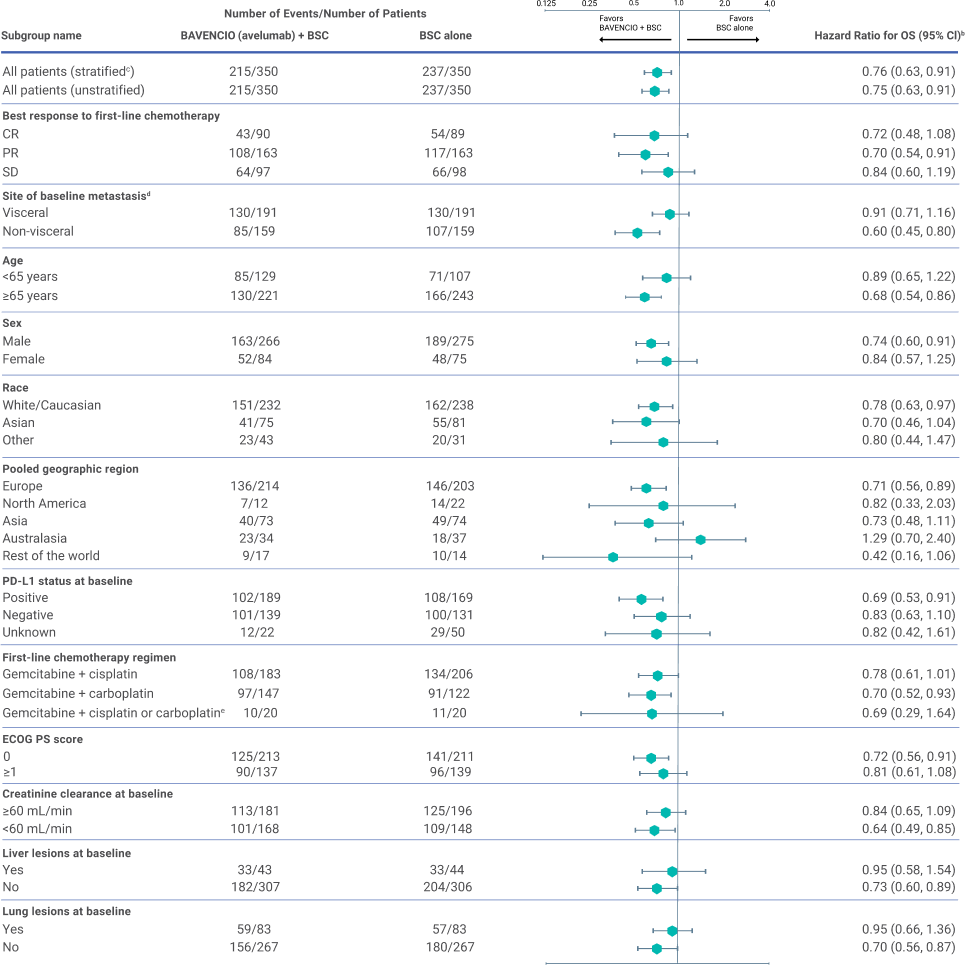
SUBGROUP ANALYSIS: OS analyses of prespecified subgroups in all randomized patients3
LIMITATIONS:
Small patient numbers can be a limitation of subgroup analyses. These results are presented for descriptive purposes and cannot be interpreted as a demonstration of efficacy in any particular subgroup. The results show the variability of the observed treatment effect over several subgroups. No adjustments were made for multiple comparisons in the subgroup analyses.
Hear from an expert on the efficacy outcomes from JAVELIN BLADDER 100 Trial
JAVELIN Bladder 100 Trial Efficacy Dr. Mehta
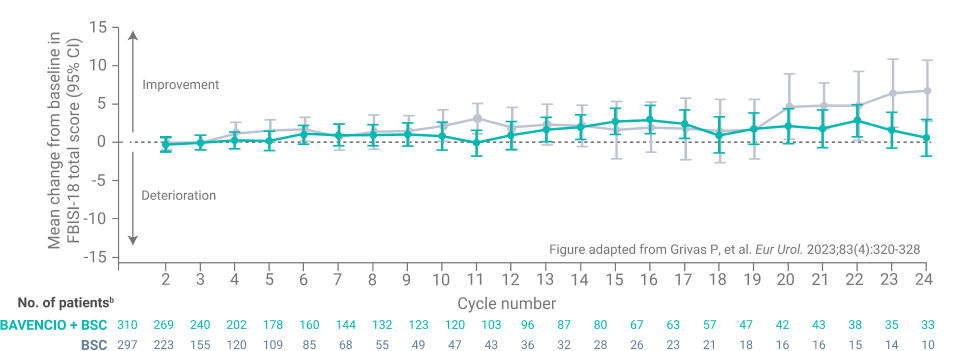
Patient-reported outcomes: A prespecified secondary endpoint from JB1009
- FBlSI-18 is a validated, bladder cancer–specific tool that measures symptoms and QOL in the past 7 days
- FBlSI-18 total score and subscales: In the mixed-effect model analysis, no significant differences were found between treatment arms in estimated mean scores or their corresponding subscales throughout all on-treatment cycles
FBlSI-18 analysis in the overall populationa
FBlSI-18 disease-related symptom subscalesa:
PHYSICAL (DRS-P)
pain, weight loss, urination, weakness, dizziness, meeting family needs, appetite, erection in males, and sleep
EMOTIONAL (DRS-E)
worrying about disease worsening, sadness
TREATMENT SIDE EFFECTS (TSE)
nausea, lack of energy, feeling ill, bowel control, bother of treatment side effects
FUNCTIONAL WELL-BEING (FWB)
ability to enjoy life, contentment with QOL
LIMITATIONS9:
- Open-label trial design and the limited number of patients providing data at later time points
- The limited number of patients at later cycles was prominent in the control arm, mainly due to progression events, which may limit the interpretation of longer-term PRO
- The FBlSI-18 instrument was validated in patients with bladder cancer, but some items may be less relevant for advanced disease in the maintenance setting
- All analyses were not adjusted for multiple testing, hindering their overall interpretation
- The methodology of this assessment does not allow conclusions based on these data
aRanges for each FBlSI-18 score: total, 0-72; DRS-P, 0-36; DRS-E, 0-8; TSE, 0-20; FWB, 0-8. Descriptive statistics were calculated for FBlSI-18 total score and subscales. Estimates of clinically important differences and changes for group comparisons: total, 3-6; DRS-P, 2-3; TSE, 1-2; and DRS-E and FWB, one each. Estimates of significant changes in individual patients: total, 3-9; DRS-P, 2-6; DRS-E, 1-3; TSE, 2-5; and FWB, 2-4.9
bNumber of patients who completed the baseline assessment and the assessment at the respective cycle. Data for on-treatment visits that had 10 or more patients in both arms are shown. For the BAVENCIO + BSC and BSC-alone arms, 333 and 330 patients responded to one or more items at baseline, respectively.9
1L=first line; BSC=best supportive care; CI=confidence interval; CR=complete response; ECOG PS=Eastern Cooperative Oncology Group Performance Status; FBISI=Functional Assessment of Cancer Therapy Bladder Symptom Index-18; HR=hazard ratio; JB100=JAVELIN Bladder 100 Trial; mOS=median overall survival; OS=overall survival; PD-L1=programmed death ligand-1; PRO=patient-reported outcome; QOL=quality of life; PR=partial response; SD=standard deviation; UC=urothelial carcinoma.
References: 1. PR Newswire. FDA Approves BAVENCIO as first-line maintenance treatment for patients with locally advanced or metastatic urothelial carcinoma. Press release. June 30, 2020. Accessed August 14, 2025. https://www.prnewswire.com/news-releases/fda-approves-bavencio-as-first-line-maintenance-treatment-for-patients-with-locally-advanced-or-metastatic-urothelial-carcinoma-301086396.html 2. Bavencio Prescribing Information. EMD Serono, Inc. 3. Powles T, Park SH, Caserta C, et al. Avelumab first-line maintenance for advanced urothelial carcinoma: results from the JAVELIN Bladder 100 Trial after ≥2 years of follow-up [supplementary appendix]. J Clin Oncol. 2023;41(19):3486-3492. 4. Grivas P, Park SH, Voog E, et al. Avelumab first-line maintenance therapy for advanced urothelial carcinoma: comprehensive clinical subgroup analyses from the JAVELIN Bladder 100 phase 3 trial. Eur Urol. 2023;84(1):95-108. 5. Data on file. EMD Serono, Inc., Boston, MA. 6. Powles T, Park SH, Voog E, et al. Avelumab maintenance therapy for advanced or metastatic urothelial carcinoma. N Engl J Med. 2020;383(13):1218-1230. 7. Sridhar SS, Powles T, Gupta S, et al. Avelumab first-line (1L) maintenance for advanced urothelial carcinoma: long-term follow-up from the JAVELIN Bladder 100 trial in subgroups defined by 1L chemotherapy regimen and analysis of overall survival from start of 1L chemotherapy. Abstract No. 508 presented at: 2023 ASCO Genitourinary Cancers Symposium; February 16-18, 2023; San Francisco, CA. 8. Grivas P, Park SH, Voog E, et al. Avelumab first-line maintenance in advanced urothelial carcinoma: conditional survival and long-term safety in patients treated for ≥1 or ≥2 years in JAVELIN Bladder 100. Poster #1975P presented at: European Society for Medical Oncology Congress 2024; September 13-17, 2024; Barcelona, Spain. 9. Grivas P, Kopyltsov E, Su P-J, et al. Patient-reported outcomes from JAVELIN Bladder 100: avelumab first-line maintenance plus best supportive care versus best supportive care alone for advanced urothelial carcinoma. Eur Urol. 2023;83(4):320-328.