- Results of real-world studies are limited by lack of randomization, which can reduce internal validity of data (ie, limit the ability to discern whether the difference between control and intervention groups is due to the intervention or due to other factors)
- Individual studies may have additional limitations such as small sample size, lack of control group, participants and investigators not being blinded, limited follow-up duration, poor quality of data collection, missing data, and exploratory nature of the data
Limitations of real-world data
- Retrospective studies may also be limited by sampling bias, recall bias, confounding bias, and changes in disease management practices
- The data presented are real-world analyses that reflect an observational analysis that may still be ongoing and do not meet the reliability and accuracy afforded by randomized controlled trials; data for patients may be missing due to patients not being fully compliant with follow-up visits or other factors
- Therefore, no conclusions should be drawn, and no treatment decisions should be made based on real-world analyses
REAL-WORLD EFFICACY AND SAFETY OF BAVENCIO
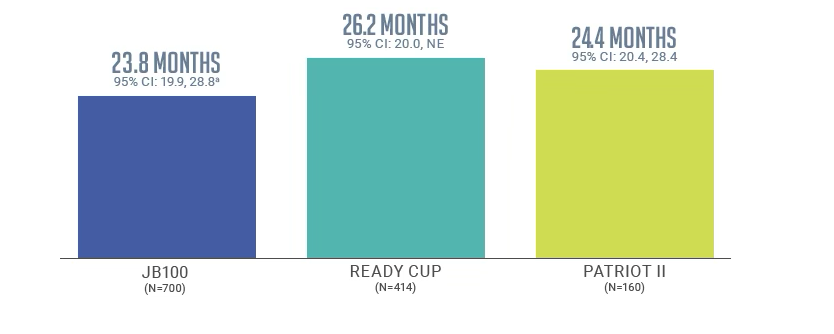
mOS from start of BAVENCIO maintenance therapy in JB100 and real-world studies1,3,4
Study Designs
READY CUP
Prospective, noninterventional, Italian, compassionate-use study involving 414 patients with la/mUC who received BAVENCIO 1L maintenance therapy (800 mg intravenously every 2 weeks). 1L treatment: 53% received a carboplatin-based regimen; 44% received a cisplatin-based regimen.3
PATRIOT II
Retrospective, observational, multicenter chart review conducted across community and academic sites in the United States. One hundred sixty patients with la/mUC were treated with BAVENCIO 1L maintenance therapy (800 mg intravenously every 2 weeks in the majority of patients [81%]).1L treatment: 38% received a carboplatin-based regimen; 62% received a cisplatin-based regimen.4,5
aBSC alone: 15.0 months mOS (95% CI: 13.5, 18.2); HR: 0.76 (95% CI: 0.63, 0.92).1
REAL-WORLD SAFETY1,3,4
Consistent safety shown across heterogeneous patient populations in everyday clinical practice3,4
Summary of safety with BAVENCIO in PATRIOT II (N=160)4
| ALL PATIENTS (N=160) | ||
|---|---|---|
| Any TRAR, n (%) | 62 (38.8) | |
| Time to onset, mean (SD), days | 95 (127) | |
| Median (range) | 56 (0-793) | |
| Any immune-related AR, n (%) | 35 (21.9) | |
| Time to onset, mean (SD), days | 146 (173) | |
| Median (range) | 91 (0-793) | |
| Therapy stopped due to any TRAR, n (%) | 16 (10.0) | |
| Therapy delayed due to any TRAR, n (%) | 20 (12.5) | |
| Received steroid (including topical) due to any TRAR, n (%) | 36 (32.1) | |
| Received high-dose systemic steroid due to any TRAR, n (%) | 23 (14.3) | |
| Outcome of TRARs, n (%) | N=165a | |
| Resolved | 105 (63.6) | |
| Unresolved | 32 (19.4) | |
| Resolved with sequelae | 2 (1.2) | |
| Unknown | 26 (15.8) | |
| Duration of TRAR(s), daysb | N=165a | |
| Mean (SD) | 97 (151) | |
| Median (range) | 31 (0-657) | |
| Hospitalized due to TRAR, n (%) | ||
| Yes | 13 (8.1) | |
| No | 147 (91.9) | |
Rates of discontinuation and other key safety measures in real-world studies are consistent with those seen in JB100.1,3,4
aN represents the number of ARs, duplicated in data file.4
bMost recent follow-up date used if unresolved.4
Hear an expert discuss the real-world evidence for BAVENCIO
Real-World Data Dr. Karimi
1L=first line; AR=adverse reaction; BSC=best supportive care; CI=confidence interval; HR=hazard ratio; JB100=JAVELIN Bladder 100; la/mUC=locally advanced or metastatic urothelial carcinoma; mOS=median overall survival; NE=not evaluable; SD=standard deviation; TRAR=treatment-related adverse reaction.
References: 1. Bavencio Prescribing Information. EMD Serono, Inc. 2. Powles T, Park SH, Caserta C, et al. Avelumab first-line maintenance for advanced urothelial carcinoma: results from the JAVELIN Bladder 100 Trial after ≥2 years of follow-up [supplementary appendix]. J Clin Oncol. 2023;41(19):3486-3492. 3. Antonuzzo L, Maruzzo M, De Giorgi U, et al. READY: Real-world data from an Italian compassionate use program of avelumab first-line maintenance for locally advanced or metastatic urothelial carcinoma. ESMO Open. 2024;5:100068. 4. Grivas P, Barata PC, Moon HH, et al. Avelumab first-line maintenance therapy for locally advanced/metastatic urothelial carcinoma: results from the real-world US PATRIOT-II study. Abstract #697 presented at: ASCO Genitourinary Cancers Symposium. January 25-27, 2024; San Francisco, CA. 5. Grivas P, Barata PC, Moon HH, et al. Baseline characteristics from a retrospective, observational, us-based, multicenter, real-world study of avelumab first-line maintenance in locally advanced/metastatic urothelial carcinoma (PATRIOT-II). Poster #465 presented at: American Society of Clinical Oncology. June 2-6, 2023; Chicago, IL.






